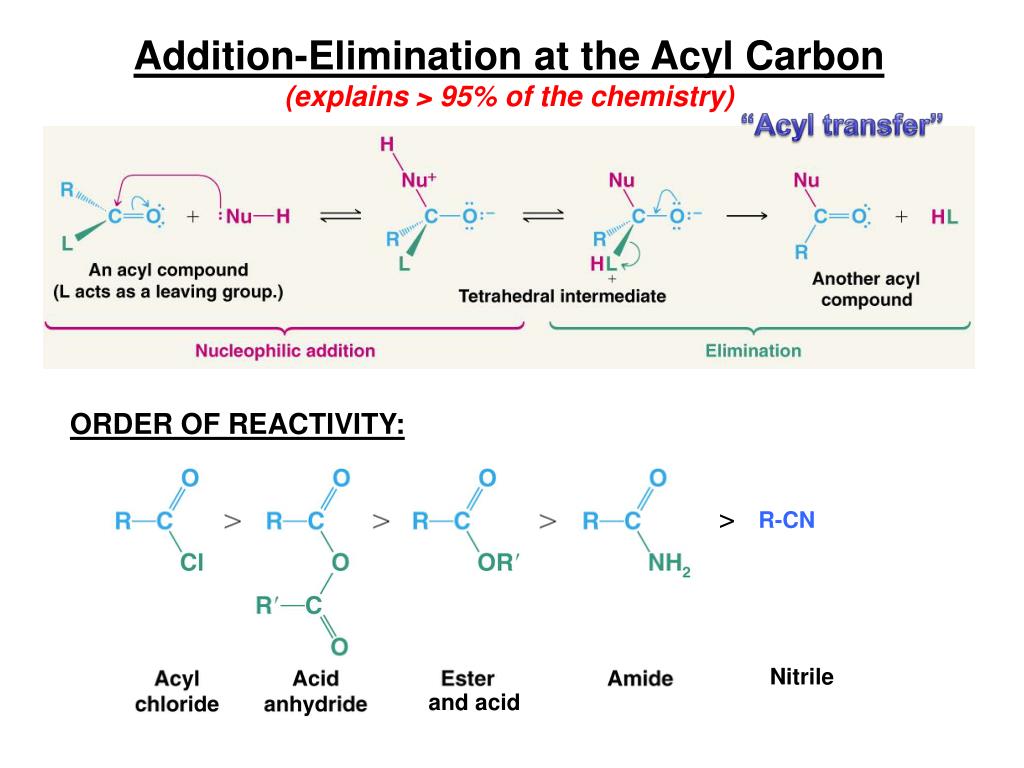
 what we're looking at for carboxylic acid derivates is the ability of the functional group to stabilize a positive charge on the carbonyl carbon by donating electrons, essentially transferring the positive charge from the carbonyl carbon to the functional group, decreasing reactivity.Īcyl halides: very poor resonance, very high reactivity. 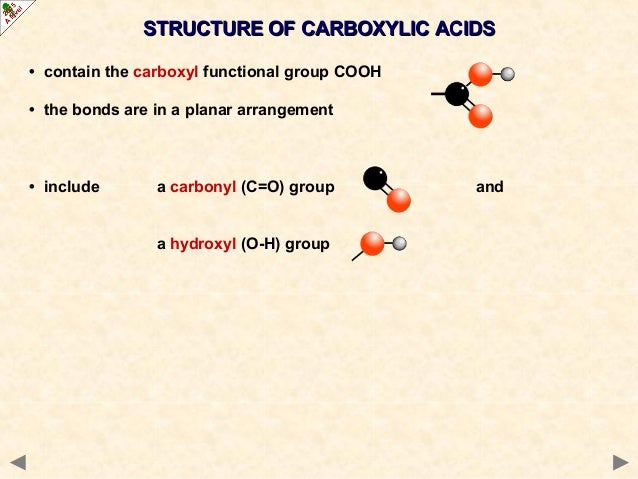
Generally an increase in resonance = an increase in stability = an decrease in reactivity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
